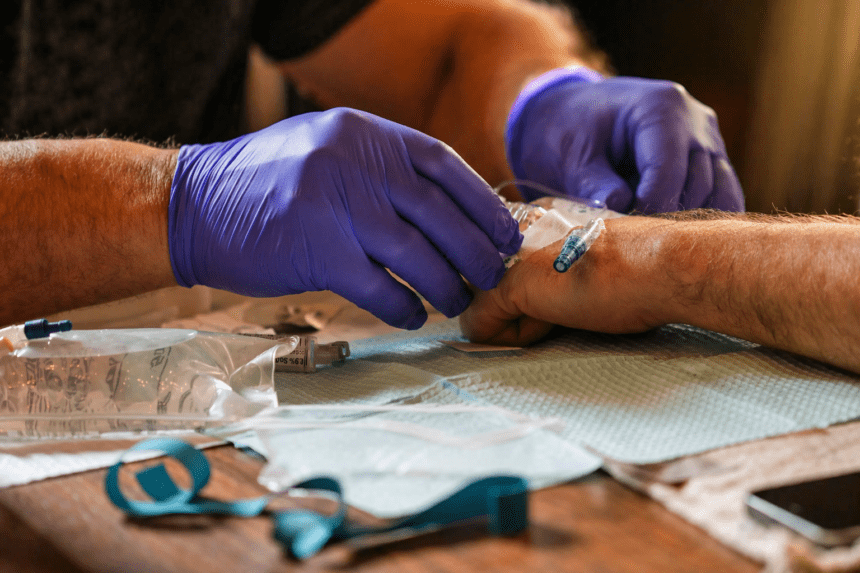
A major new analysis has thrown the Alzheimer’s field back into argument, just as two long-awaited drugs were beginning to look like the first real crack in decades of failure. The review, published by Cochrane on April 16, says anti-amyloid antibodies probably make little or no clinically meaningful difference for people with mild cognitive impairment or mild dementia caused by Alzheimer’s disease, even though they do reduce amyloid in the brain. It examined 17 trials involving 20,342 participants and also pointed to higher risks of brain swelling and bleeding.
That is the line that landed hardest: not that the drugs do nothing, but that whatever benefit they offer may be too small for patients and families to truly feel in day-to-day life. For a disease with no cure and a history of shattered hopes, that distinction matters. A treatment can clear a biological target, post a statistically significant result, and still leave doctors arguing over whether it changes enough to justify the burden.
The drugs at the center of the storm are lecanemab, sold as Leqembi, and donanemab, sold as Kisunla. Both are approved in the United States for people in the early stages of Alzheimer’s disease — specifically those with mild cognitive impairment or mild dementia and confirmed amyloid pathology. Their labels also carry prominent warnings about ARIA, the shorthand for amyloid-related imaging abnormalities, which can involve swelling or bleeding in the brain.
Supporters of the medicines were quick to push back. Their main complaint is pretty straightforward: the Cochrane review pooled older failed antibodies with the newer drugs that actually cleared late-stage trials and won approval. On that view, the review is not exactly wrong, but it muddies the picture by treating one uneven drug class as though all the molecules had performed the same way. Several experts quoted by the UK’s Science Media Centre made that argument directly, saying the conclusions are weakened by combining five unsuccessful drugs with two that did slow decline, albeit modestly.
And there is evidence behind that objection. In the phase 3 CLARITY AD trial, lecanemab slowed decline on the Clinical Dementia Rating–Sum of Boxes compared with placebo over 18 months. Donanemab’s TRAILBLAZER-ALZ 2 trial also reported significantly less decline than placebo in people with early symptomatic Alzheimer’s disease. Those results were never marketed by serious researchers as a miracle. Still, they were enough to convince regulators that the drugs had crossed a threshold older amyloid therapies never reached.
Even so, the critics of the drugs say the new review cuts through years of hype. One expert reaction published Thursday described the average treatment effect seen in trials as roughly 2% of the scale range on standard measures of cognition or function — statistically detectable, yes, but unlikely to amount to an obvious difference for an individual patient sitting in a clinic room. That camp argues the conversation has often drifted from “modest slowing” into something far more hopeful than the science really supports.
That leaves patients and families in a hard place. Because here’s the truth: both sides are talking about something real. The newer drugs do appear to slow decline in selected early-stage patients. At the same time, they require regular infusions, monitoring, scans, and careful screening for side effects. They are not simple drugs to take, and they are not risk-free. So the question stops being “Do they work?” and becomes more personal, and more difficult: “Do they work enough?”
Health systems are answering that question differently. In the UK, NICE said in its June 2025 final draft guidance that the benefits of donanemab and lecanemab remained too small to justify the extra cost to the NHS. The agency said the treatments had been shown to delay progression from mild to moderate Alzheimer’s by about four to six months, but concluded that the gain was still not enough, given the price of the drugs, their administration, and the wider pressure on NHS resources. NICE is revisiting the issue after further consultation, which shows the matter is far from settled.
There is another layer to this debate that often gets lost in the headlines. Alzheimer’s researchers are increasingly talking less like true believers in the old amyloid theory and more like people trying to build a more complicated map of the disease. Even experts defending the approved antibodies now say amyloid removal is probably not the whole answer. Genetics, inflammation, vascular changes, and timing may all shape whether a patient gets any measurable benefit. In other words, the field may be inching forward, but it is still inching.
So this week’s row is not really about whether anti-amyloid drugs are a fraud or a breakthrough. It is about standards. How much slowing counts as meaningful? How much risk is acceptable? How much should health systems pay for a few extra months before decline deepens? Those aren’t laboratory questions anymore. They are policy questions, family questions, and, frankly, moral questions. The review has stirred the debate because it forces the field to say out loud what it has long known privately: after all the excitement, Alzheimer’s treatment has moved forward — but not by much.