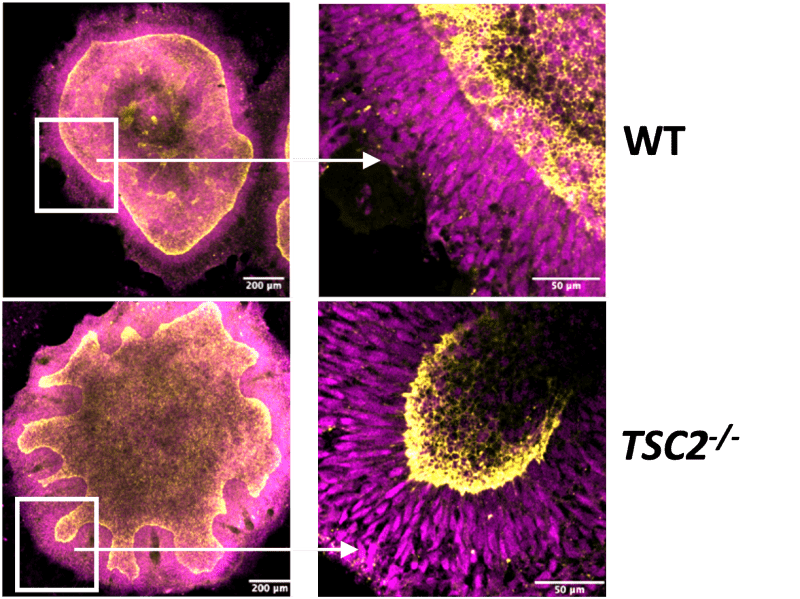
3D bioprinting is rapidly transforming medical research, allowing scientists to create complex organoids and functional tissues with unprecedented precision. This technology promises to revolutionize drug development, disease modeling, and, eventually, regenerative medicine.
The ability to print miniature organs and tissue structures outside the body offers a critical alternative to animal testing, providing more accurate disease models for drug efficacy and toxicity studies. It also holds the long-term potential to address the critical shortage of donor organs for transplant patients. Researchers worldwide are now routinely bioprinting everything from liver organoids to cardiac patches and kidney structures.
These engineered tissues mimic human biology more closely than traditional 2D cell cultures, offering a vital window into disease progression and treatment responses.
The precision offered by 3D bioprinting allows for a level of control over cellular architecture that was previously unimaginable, according to experts in the field. Yet, significant challenges persist. Integrating a functional vascular system a network of blood vessels into larger bioprinted tissues remains a major hurdle.
Without it, cells deep within the structure cannot receive adequate nutrients or remove waste, limiting tissue viability and size. Selecting the right “bio-inks” biocompatible materials that can be printed and support cell growth also requires continuous innovation. Despite these obstacles, the trajectory of 3D bioprinting points towards a future where personalized medicine and regenerative therapies are fundamentally reshaped.
“This technology is moving us closer to understanding complex human diseases in ways we couldn’t before,” one lead researcher noted recently. The ongoing advancements suggest a future where patient-specific tissues could be printed on demand, moving science beyond theoretical models into tangible, functional biological solutions.