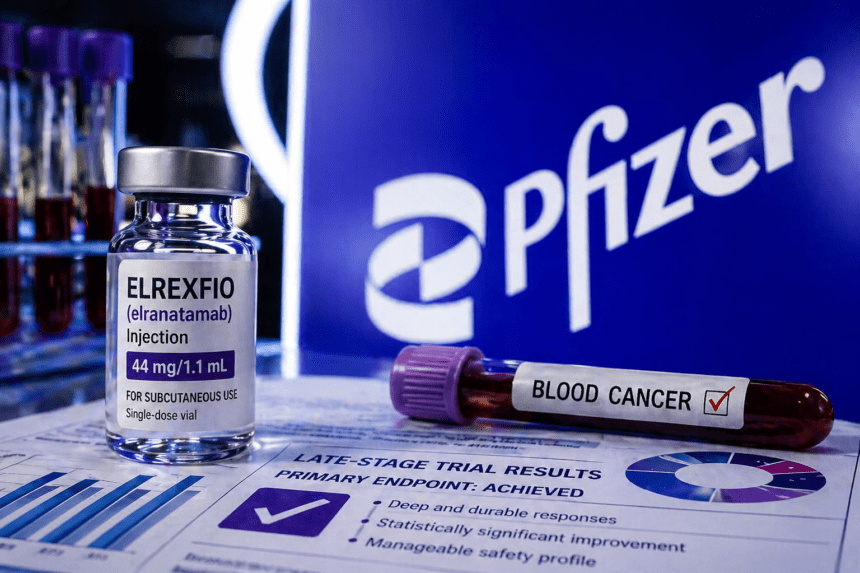Pfizer’s immunotherapy, Elrexfio, has met its primary objective in a late-stage clinical trial, offering a potential new path for patients battling relapsed or refractory multiple myeloma. Data released by the pharmaceutical giant shows the drug successfully extended progression-free survival in patients who had previously exhausted standard treatment options.
The drug, a B-cell maturation antigen (BCMA)-directed CD3 T-cell engager, works by forcing the immune system to hunt down and destroy cancer cells. In this specific trial, patients receiving Elrexfio saw a measurable delay in their disease worsening compared to those on conventional therapy regimens. For the medical community, these results provide more than just a statistical win.
Multiple myeloma remains a notoriously difficult cancer to manage, as patients often develop resistance to initial lines of therapy. Elrexfio’s ability to step in after those failures provides a critical safety net for a patient population with few remaining choices.
“Patients with relapsed or refractory multiple myeloma face a cycle of treatment and recurrence that is physically and mentally exhausting,” said a lead investigator involved in the study. “Seeing this level of efficacy in a late-stage setting suggests we are moving closer to making long-term management a reality rather than an exception.
Despite the positive outcome, the drug comes with a known profile of side effects. Regulators have previously flagged cytokine release syndrome—an intense immune response—as a primary safety concern. Pfizer’s latest data indicates that while these events occur, they are largely manageable through established clinical protocols, allowing patients to stay on the treatment longer.
The company is now preparing to submit this updated data to global health authorities to expand the drug’s label. If approved, it cements Elrexfio’s role as a staple in hematology wards, moving it from a last-resort option to a more standard secondary intervention.
While the pharmaceutical landscape is crowded with similar BCMA-targeting therapies, Pfizer is betting that the drug’s administration schedule and proven efficacy in this trial will give it a distinct edge. For the thousands of patients currently cycling through treatments, the trial results offer a clear, if cautious, reason for optimism.